|
Organisations of any size are applicable for ISO 13485 requirement. The purpose of these regulatory requirements is to ensure that electronic manufacturers can consistently deliver medical devices that are safe and fit for their intended purpose. 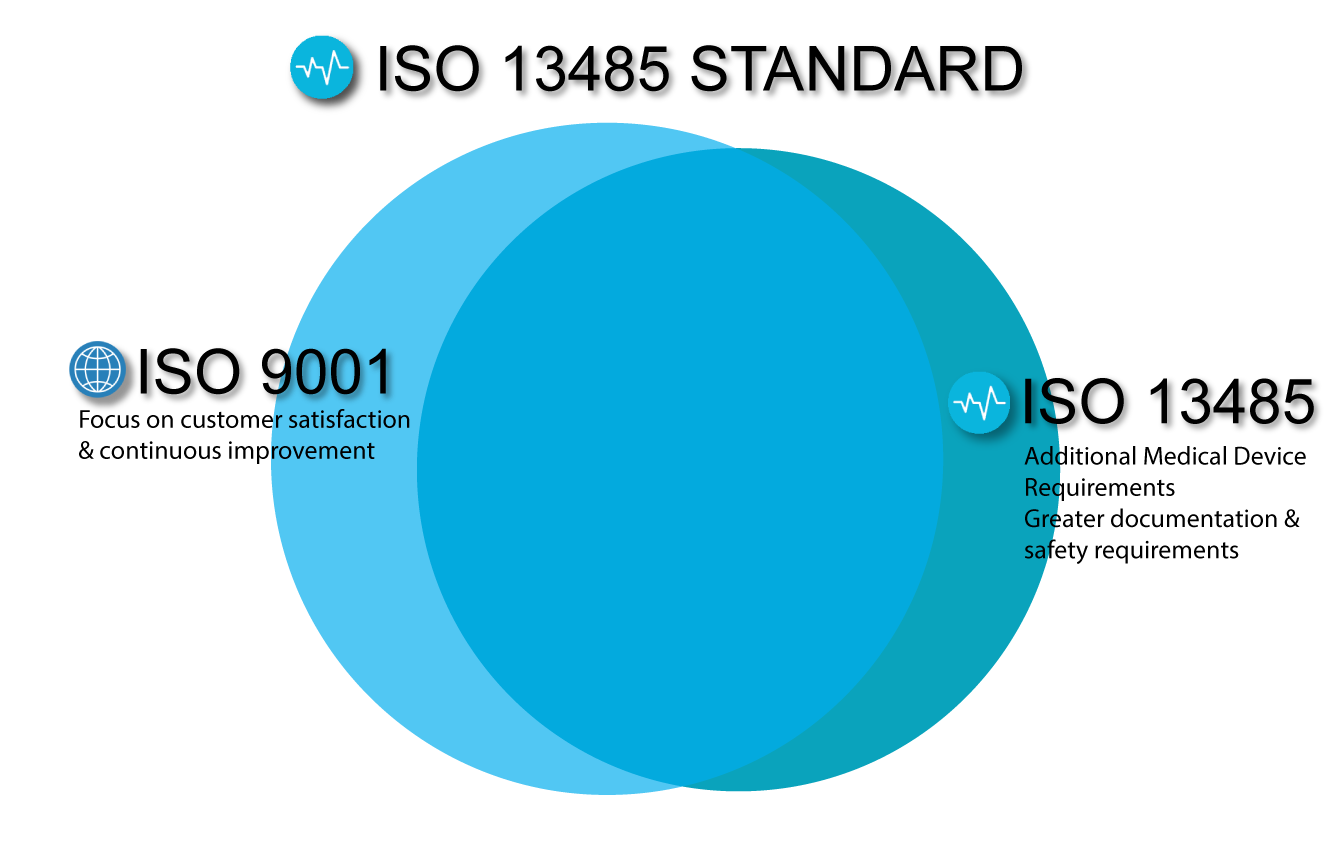
If any of the requirements are not met or the registrations procedures are not completed, placing a product on the market is impossible. The manufacturing of medical devices is one of the most regulated sectors in which many regulations and obligations must be achieved.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
